Introduction
The AMARIS product family has a new addition. The SCHWIND AMARIS 1050RS is the new flagship, which is among the leading excimer laser technologies today. It is the only excimer laser system available in the market to operate at 1050 Hz, leading to an extremely short ablation time of just 1.3 seconds per dioptre1.
Another innovative feature of the SCHWIND AMARIS 1050RS is active 7D eye tracking in space and time. With this feature, eye movements occurring during the period between acquisition of the eye-tracker image and triggering of the subsequent laser pulses are anticipated and pre-compensated.
This results in zero latency time for the laser system, which means even greater safety during laser treatment. A recent study proves that results obtained with SCHWIND AMARIS 1050 RS are very precise and predictable.
Results
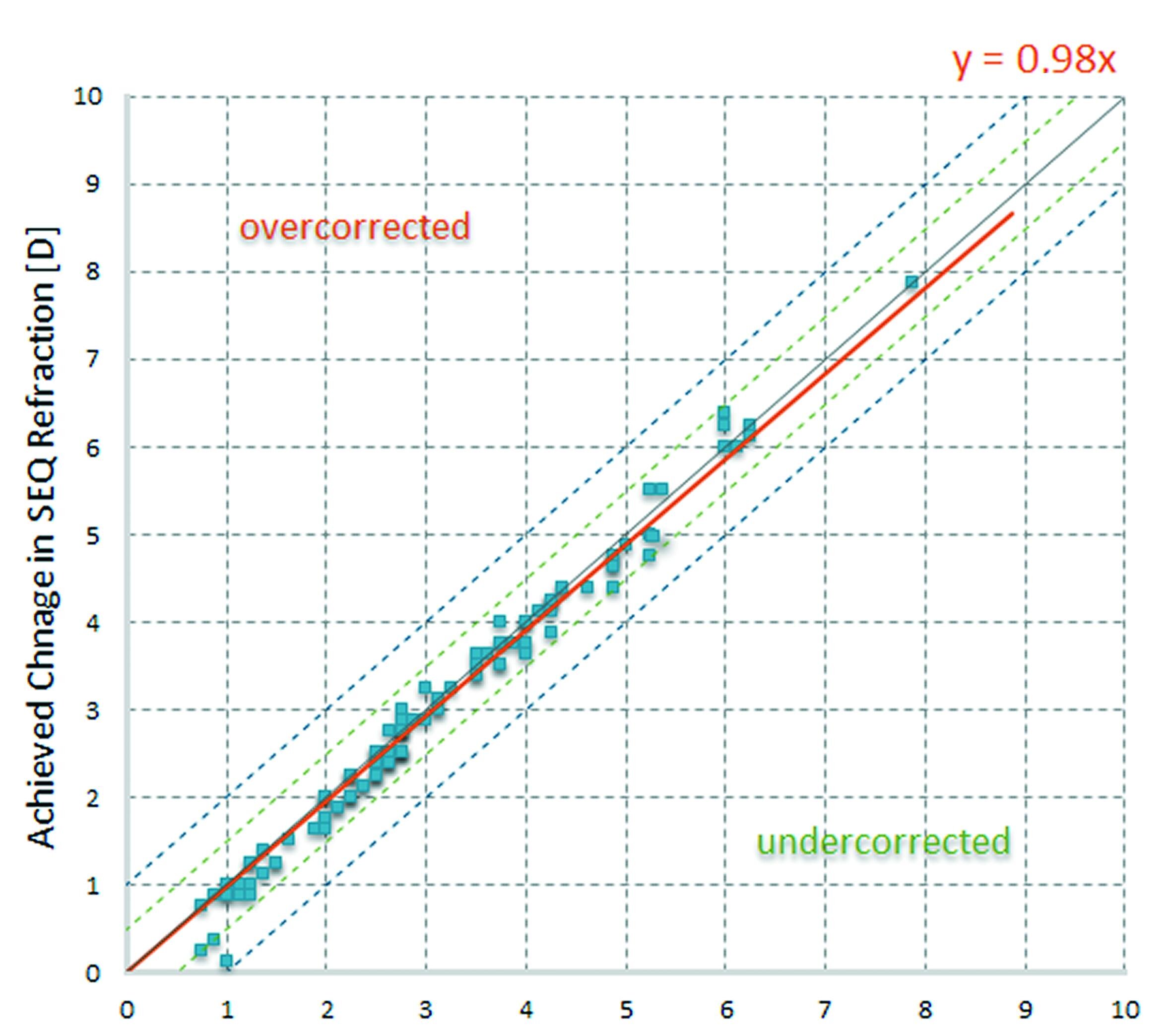
Scattergram
Figure 1 shows attempted refraction [D] vs. achieved refractive change for each single eye at three months postoperative.
The narrow scatter between attempted and achieved change in SEQ (spherical equivalent) at three months is obvious. The mean postoperative SEQ was +0.02 D ±0.27 D.
Refractive outcome
Figure 2 displays the refractive outcome in terms of spherical equivalent (SEQ). An impressive 54% of all 114 evaluated eyes were in the range of ± 0.125 D and 99% were within ±0.50 D in terms of SEQ.
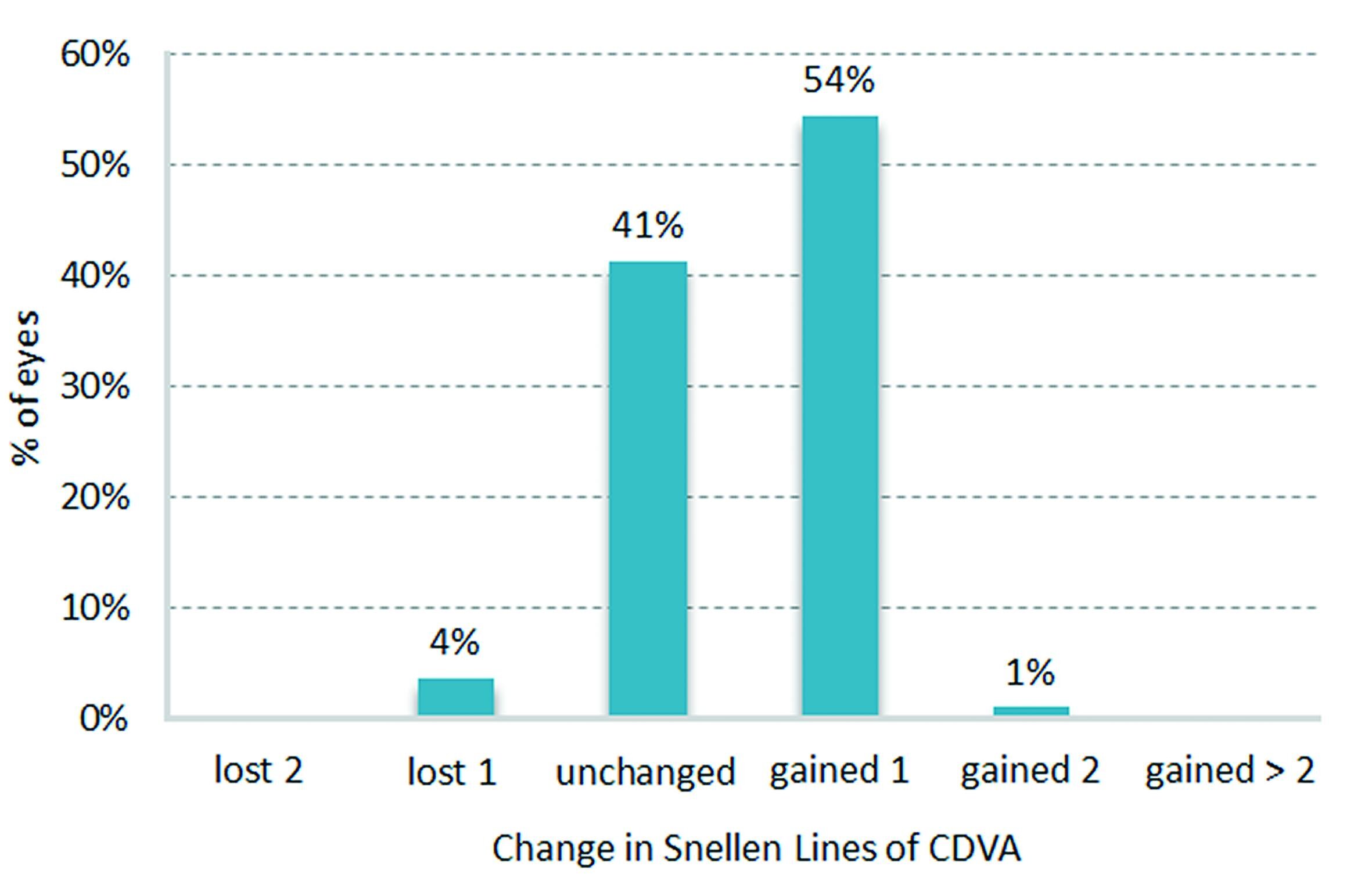
Change in BSCVA-safety
Safety is described by the change in best spectacle corrected visual acuity (BSCVA) measured by the gain or loss of Snellen lines from preoperative to postoperative. At three months, 55% of all evaluated eyes gained one or more Snellen lines. No patient lost more than one Snellen line in terms of BSCVA (see figure 3).
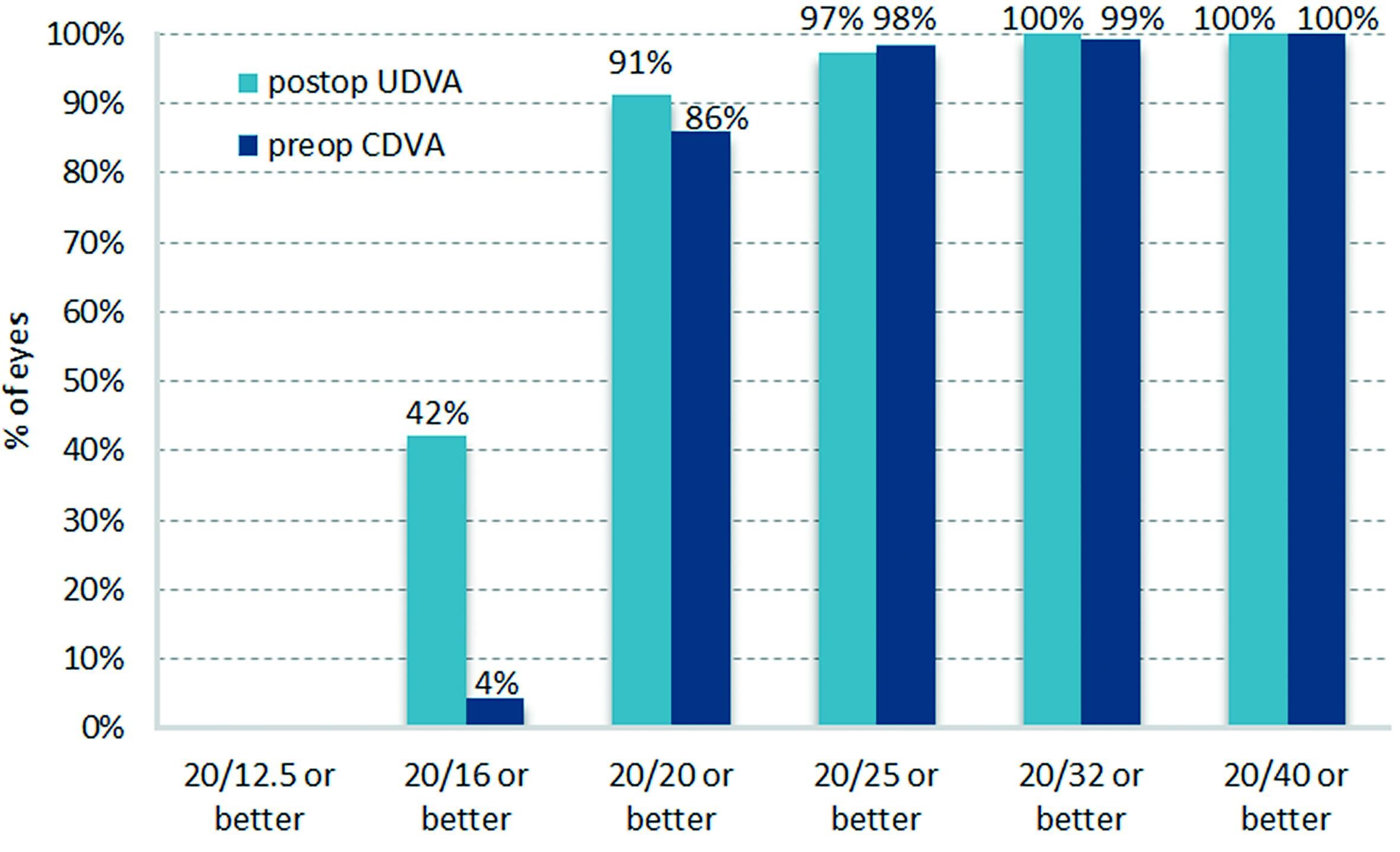
Preop. BSCVA versus postop. UCVA
Figure 4 displays the preoperative best spectacle corrected visual acuity (BSCVA) vs. the postoperative uncorrected visual acuity (UCVA) at three months. It shows that postoperatively 42% of all treated eyes have an uncorrected visual acuity (UCVA) of 20/16 or better.
In summary a significant change towards an improved visual acuity is clearly obvious.
Astigmatism
Figure 5 shows the refractive astigmatism preoperatively and three months postoperatively.
The mean postoperative cylinder was -0.10 D ±0.19 D. In general, a significant reduction in refractive astigmatism is obvious. Postoperatively, 96% of all treated eyes have a refractive astigmatism of equal to or smaller than 0.25 D. All eyes are within +/- 0.5 D of astigmatism.
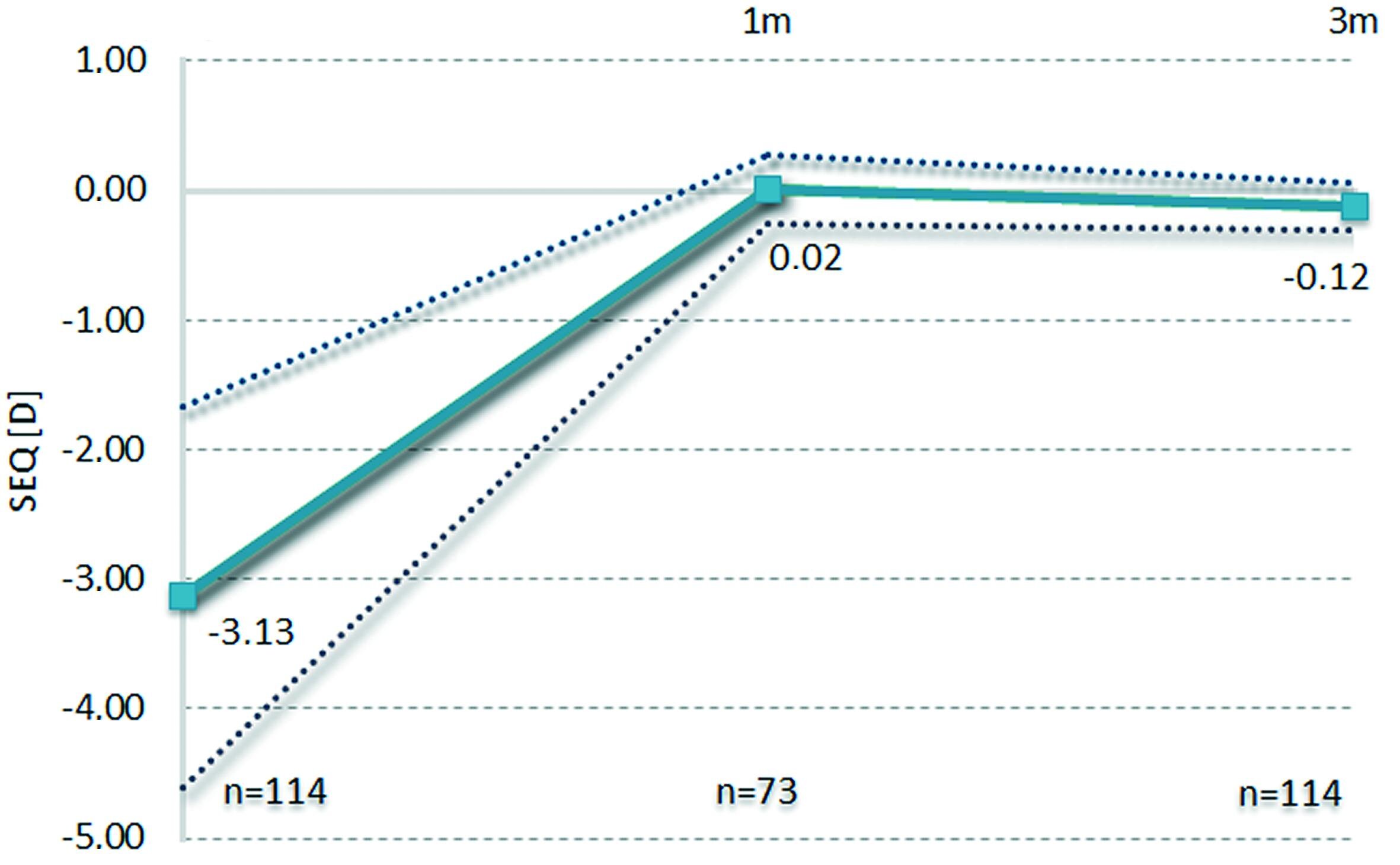
Achieved correction over time – stability
The stability of the results for the follow up examinations of one month and three months in figure 6 shows the highly successful rate.
Aberrations
When a patient is selected for non-customised aspherical treatment, the global aim of the surgeon should be to leave any existing higher order aberrations (HO) unchanged because the best corrected visual acuity, in such patients, is unaffected by the pre-existing aberrations. Hence, all factors that may induce HO, such as biomechanics, need to be taken into account prior to the treatment to ensure that the preoperative HO are unchanged after treatment.
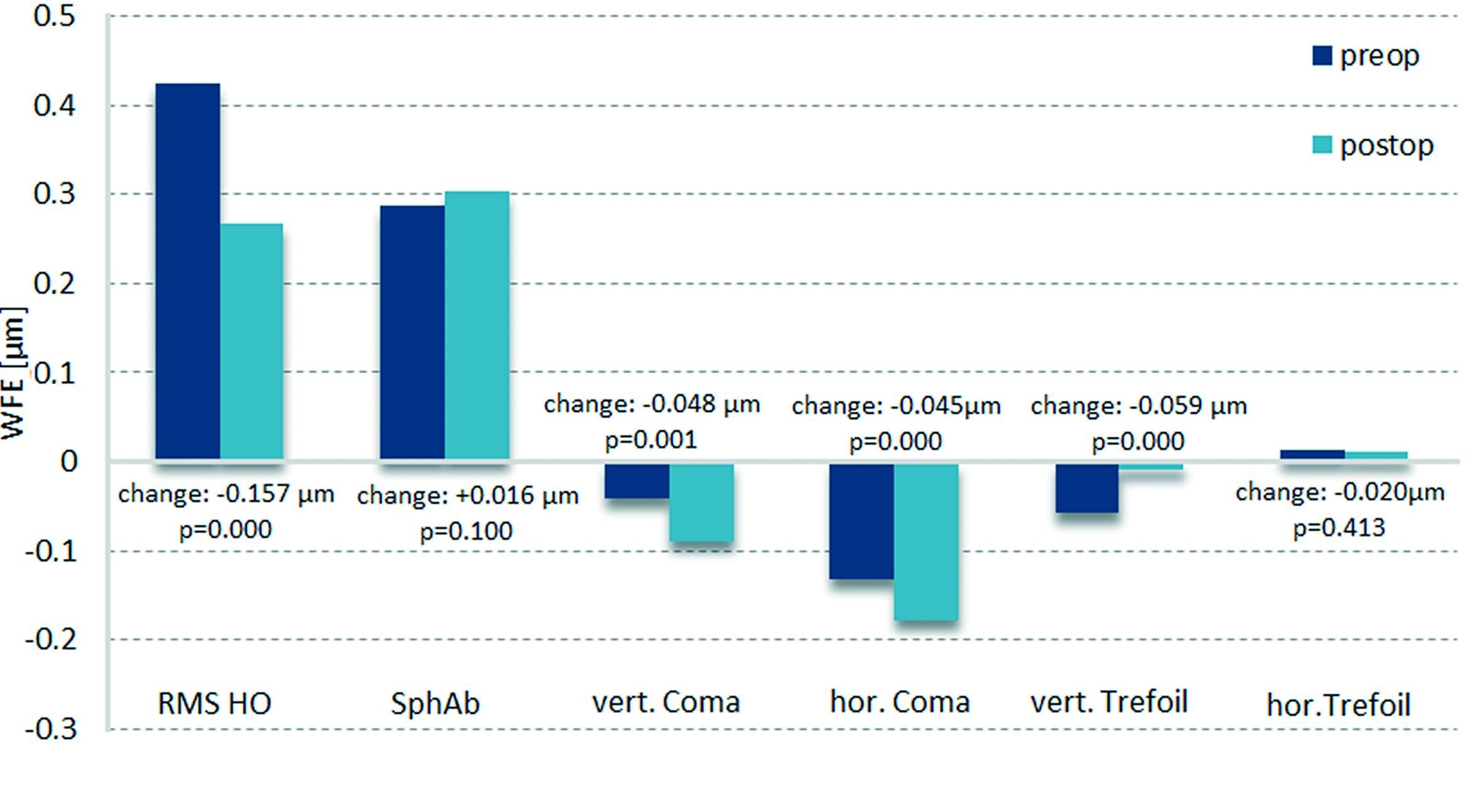
The change in spherical aberration, horizontal and vertical coma, horizontal and vertical trefoil and average value for higher order aberrations (RMS HO) was analysed at a zone of 6 mm for all eyes in which pre- and postoperative topographies were available (see figure 7).
The mean change from preoperative to three months was +0.016 µm for spherical aberration (p = 0.10), -0.048 µm for vertical coma (p<0.05), -0.045 µm for horizontal coma (p<0.05), +0.059 µm for vertical trefoil (p<0.05), -0.020 µm for horizontal trefoil (p = 0.413) and -0.157 µm for RMS HO (p<0.05). The change in spherical aberration and horizontal trefoil appeared to be not statistically significant. RMS HO was reduced slightly, although targeting for no change in higher order aberrations.
As far as our experience goes, the amount of induced corneal spherical aberration in“Aberration-free” LASIK is clearly lower compared to other published articles. In wavefront-guided ablation, the objective is not only to reduce the induction but also to reduce the aberration of the eye. In the case of aspheric “Aberration-free” treatment the aim is to remodel the slope of the cornea to compensate for the attempted sphere and cylinder components, without inducing new aberrations.
In summary the results of the aberration analysis prove the minimum induction of aberrations with “Aberration-free” profile.
1 Myopia, without astigmatism, 12.5 mm vertex distance and 6 mm optical zone
![Refractive outcome within attempted SEQ [D], 114 eyes Refractive outcome within attempted SEQ [D] in a study of 114 eyes](/infocenter/newsletter/studien/2013-1050rs-lasik-6m-results/407/image-thumb__407__teaser_image/refractive-outcome-within-attempted-seq-114eyes.4706949a.jpg)
![Refractive astigmatism [D] preoperative and three months postoperative, 114 eyes Refractive astigmatism [D] preoperative and three months postoperative in a study of 114 eyes](/infocenter/newsletter/studien/2013-1050rs-lasik-6m-results/406/image-thumb__406__teaser_image/refractive-astigmatism-preoperative-and-three-months-postoperative-114eyes.0162a146.jpg)