Many of us come across patients who are dissatisfied with their visual outcome following treatment, despite an uncorrected postoperative visual acuity of 20/20 without residual refraction. The cause of their dissatisfaction can be postoperative corneal residual astigmatism, even if this does not manifest in the refraction. A study documents that, in seven percent of all cases when exclusively refractive parameters (manifest, cycloplegic, wavefront) are used in the treatment plan, without considering corneal astigmatism, there is greater corneal astigmatism postoperatively than preoperatively.1
When planning refractive treatments particularly for patients with astigmatism, it is very important to calculate the ocular residual astigmatism (ORA) preoperatively. This is defined as the vectorial difference between the corneal astigmatism and the refractive cylinder (VD=0) and is expressed in diopters. A previously cited study shows that about one-third of the eyes have an ORA greater than 1.0 D pre-operatively.1 In such cases with high ORA (>1.00 D), the surgeon should plan the cylinder correction particularly carefully to avoid a high degree of postoperative corneal astigmatism.
With Vector Planning, I have developed a method which systematically incorporates corneal parameters in the refractive treatment plan. This results in a more natural corneal shape with fewer aberrations and a reduction in postoperative symptoms, such as glare or halos. Treatments based exclusively on refractive parameters aim to neutralise the ORA on the cornea. In terms of astigmatism correction, this approach leads in many cases to a worse result than preoperatively measured. In the case of treatments based solely on corneal parameters (topography-guided), the total extent of the ORA remains in the refraction, and in turn does not lead to optimal cylinder correction.
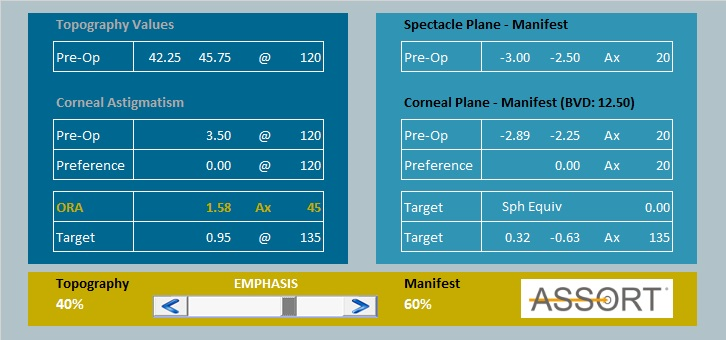
Studies with vector planning2,3 have shown that an optimal effect is achieved with a weighting of the refractive astigmatism from 50 to 65 percent and of the corneal astigmatism from 35 to 50 percent of the ORA. As an example, let us take Figure 1, which is based on a manifest refraction of -3.00/-2.50 Ax 20 and a corneal astigmatism with topographically simulated keratometry values of 42.25 @ 30/45.75 @ 120. This results in an ORA of 1.58D Ax 45. With a 60 percent weighting of the ORA by refractive astigmatism, the targeted refractive cylinder is -0.63D Ax 135, and correspondingly the corneal cylinder is 0.95D @ 135.
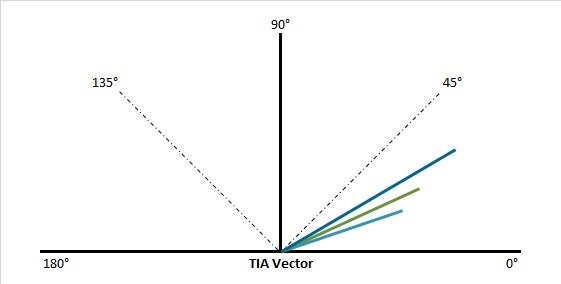
The astigmatism treatment is determined according to the planned cylin-der correction. This is defined via the vectorial difference between the preoperative parameters (VD=0) and the intended astigmatism correction (Fig. 2). Half of the intended refractive cylinder should be compensated for by a spherical component in order to achieve a spherical equivalent of zero (0.32/-0.63 Ax 135). This compensation is highly appreciated by the patients. If the treatment is based on 100 percent of the manifest refraction, the ORA of 1.58 D in our example, is neutralised on the cornea postoperatively. If the treatment is based 100 percent on corneal parameters, ocular residual astigmatism is seen in the refraction. For the purpose of vector planning (VD=0), the target value (0.32/-0.63 Ax 135) is subtracted from the manifest refraction (-2.89/-2.25 Ax 20) to give the following values: -2.66/-2.71 Ax 25. The result is a reduction in post-operative corneal residual astigmatism with a simultaneous decrease in higher-order aberrations, a reduction in refractive cylinder in practice due to the better corneal shape and the potential to improve the best corrected visual acuity.
References
1 Alpins NA. New method of targeting vectors to treat astigmatism. J Cataract Refract Surg 1997; 23:65-75
2 Alpins NA, Stamatelatos G. Customized photoastigmatic refractive keratectomy using combined topographic and refractive data for myopia and astigmatism in eyes with forme fruste and mild keratoconus. J Cataract Refract Surg 2007; 33:591-602
3 Alpins NA, Stamatelatos G. Clinical Out-comes for laser in situ keratomileusis using combined topography and refractive wavefront treatments for myopic astigmatism. J Cataract Refract Surg 2008; 34:1250-1259